This is a 7 point assignment
Objectives: After completing this unit, students should be able to:
- Apply the Law of Conservation of Mass.
- State the basic postulates of Dalton’s Atomic Theory.
- Describe the studies made by Thomson, Millikan and Rutherford which helped develop our understanding of the subatomic nature of matter.
- Describe the composition of the atom in terms of the protons, neutrons, and electrons and the properties of these subatomic particles.
- Write the nuclear symbol of an isotope or, given the nuclear symbol, state the mass number, atomic number, and number of protons and neutrons.
- Given the atomic masses and the relative abundance of all the naturally occurring isotopes of an element, calculate the average atomic mass in amu.
- Given a periodic table, identify elements according to the following: metals, nonmetals, transition metals, halogens, noble gases, alkali metals, alkaline earth metals, periods, families and groups.
- Given the chemical formula of any atom, ion or compound listed in the “Formula and Nomenclature” handout, give the name and vice versa.
- Identify the elements that exist as diatomic molecules.
- Given a molecular formula, indicate how many atoms of a given element are in the molecule.
Reading and Homework Exercises
Table of Contents from: OpenStax Chemistry 2e: Chapter 2 – Atoms, Molecules, and Ions
Introduction
Section 2.1 Early Ideas in Atomic Theory
Section 2.1 Practice – Questions 1-4 check your knowledge of early laws and theories that helped lead chemists to today’s understanding of the atom as the basic building block of matter
Question 1:
Question 2:
Question 3:
Question 4:
Section 2.2 Evolution of Atomic Theory
Section 2.2 Practice – Questions 5-7 test your understanding of early experiments that moved scientists’ knowledge beyond the atom to a subatomic level, and the conclusions and discoveries drawn from these experiments
Question 5:
Question 6:
Question 7:
Section 2.3 Atomic Structure and Symbolism
Section 2.3 Practice – Questions 8-13 check your understanding of atomic symbols, isotopes, atomic and mass numbers, and numbers of subatomic particles
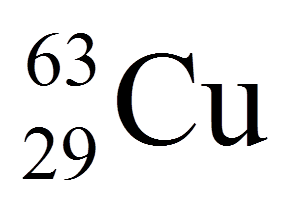
Question 8:
Question 9:
Question 10:
Question 11:
Question 12:
Question 13:
Section 2.4 Chemical Formulas
Section 2.4 Practice – Questions 14-16 test your understanding of chemical formulas
See Figure 20.4 from the Open Stax Chemistry 2e text:
Question 14:
Question 15:
Question 16:
Section 2.5 The Periodic Table
Section 2.5 Practice – Questions 17-20 cover the periodic table, periods, families (groups), and properties of different groups
Question 17:
Question 18:
Question 19:
Question 20:
Section 2.6 Molecular and Ionic Compounds
Section 2.6 Practice – Questions 21 and 22 cover molecular and ionic compounds. You should be able to distinguish which of these types a compound is by its chemical formula.
Question 21:
Question 22:
Section 2.7 Chemical Nomenclature
In addition to Section 2.7 from the OpenStax Text, see the CHEM 151 Formulas and Nomenclature Handout. It can be found on the course D2L site. Click on Content, CHEM 151 Formulas and Nomenclature – Handout, Interactive Practice, and Drills, and CHEM 151 Formulas and Nomenclature. A hard copy was also provided in your class.
Section 2.7 Practice
For practice questions, please see the interactive questions at the following link:
Interactive Nomenclature Practice:http://express.lcc.edu/nomenclature/Chem151/welcome.asp